
Submit. Faster.
Regulos generates medical device certification documents for the FDA. From source ingestion to compliance-checked submissions - in a fraction of the time.

Regulos generates medical device certification documents for the FDA. From source ingestion to compliance-checked submissions - in a fraction of the time.
What We Do
Preparing FDA submissions like eSTARs is a time-consuming, manual process filled with repetitive data entry, cross-referencing, and compliance guesswork. Regulos changes that.
Our AI ingests your source documents - test reports, clinical data, device specs - and generates submission-ready regulatory content grounded in your evidence, with precise citations and built-in compliance validation.
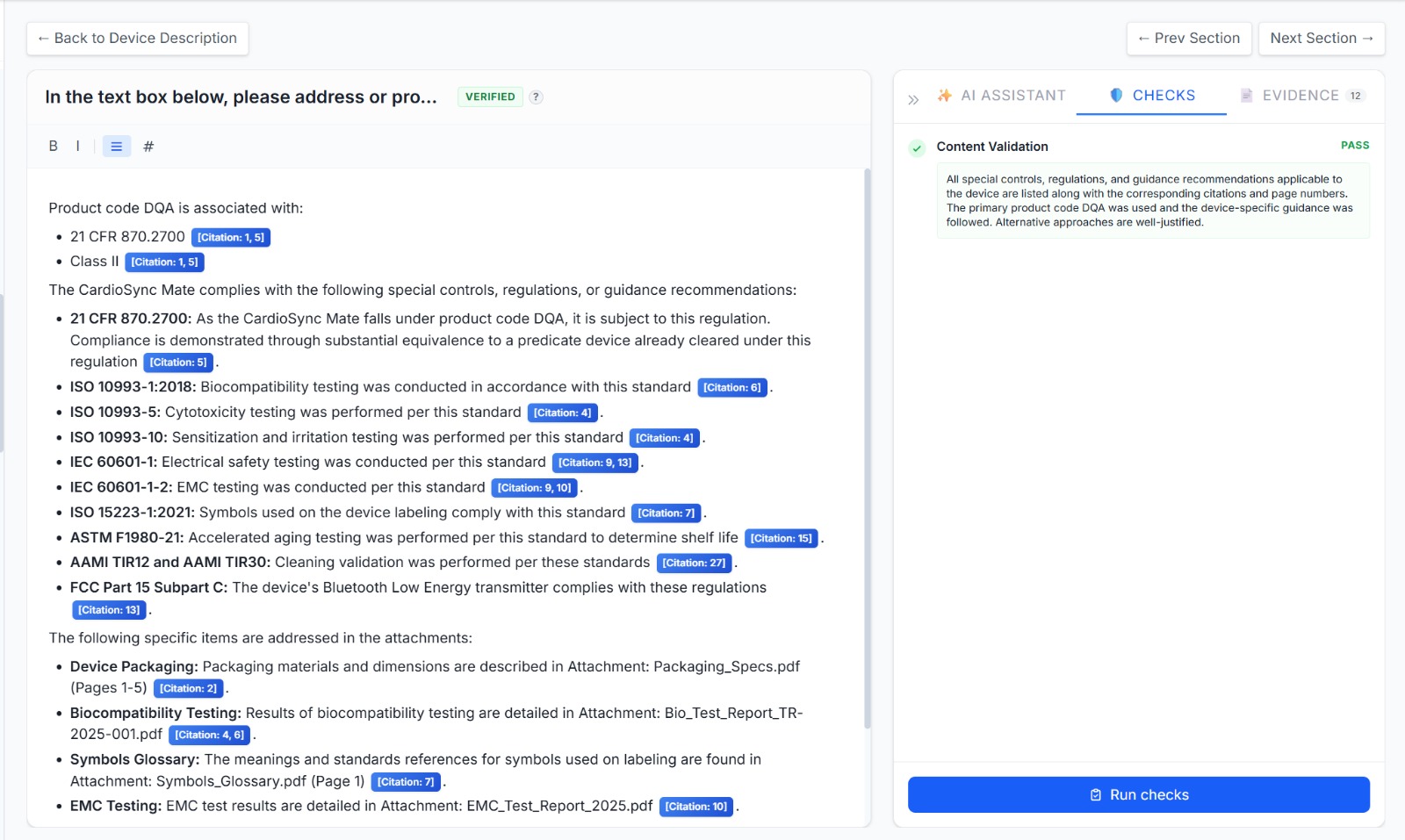
How It Works
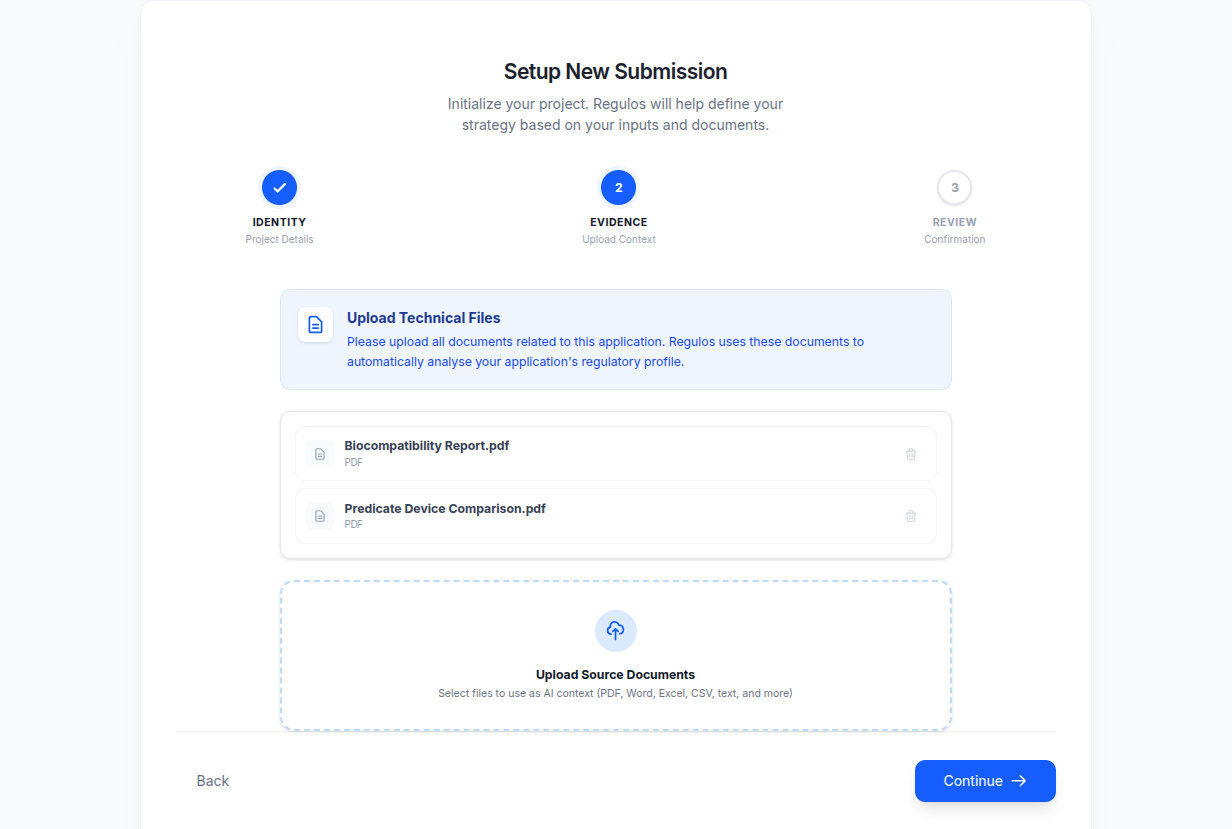
Drag-and-drop your test reports, clinical evaluations, device specifications, and reference standards. Regulos ingests, chunks, and indexes everything automatically.
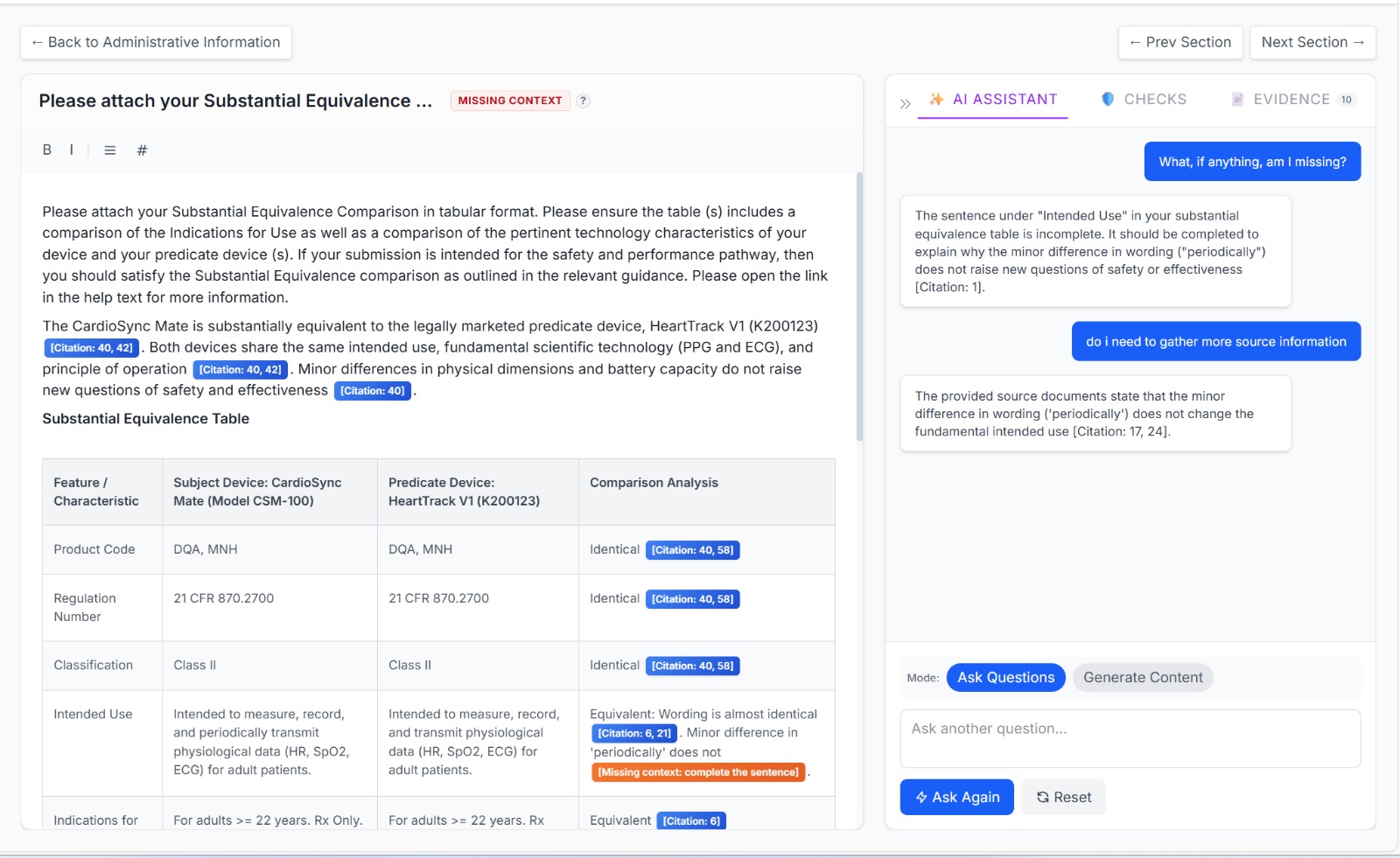
Our AI drafts each section of your eSTAR using RAG - every claim is grounded in your source evidence with inline citations.

Built-in compliance checks run continuously - regex rules, required headers, and LLM-powered validators ensure your content meets FDA expectations before you submit.

Export your completed submission to the eStar format ready to be sent to the FDA.
Ready to move faster?
Join the teams using Regulos to cut their FDA submission timeline from months to days. Reach out and we'll get you set up.